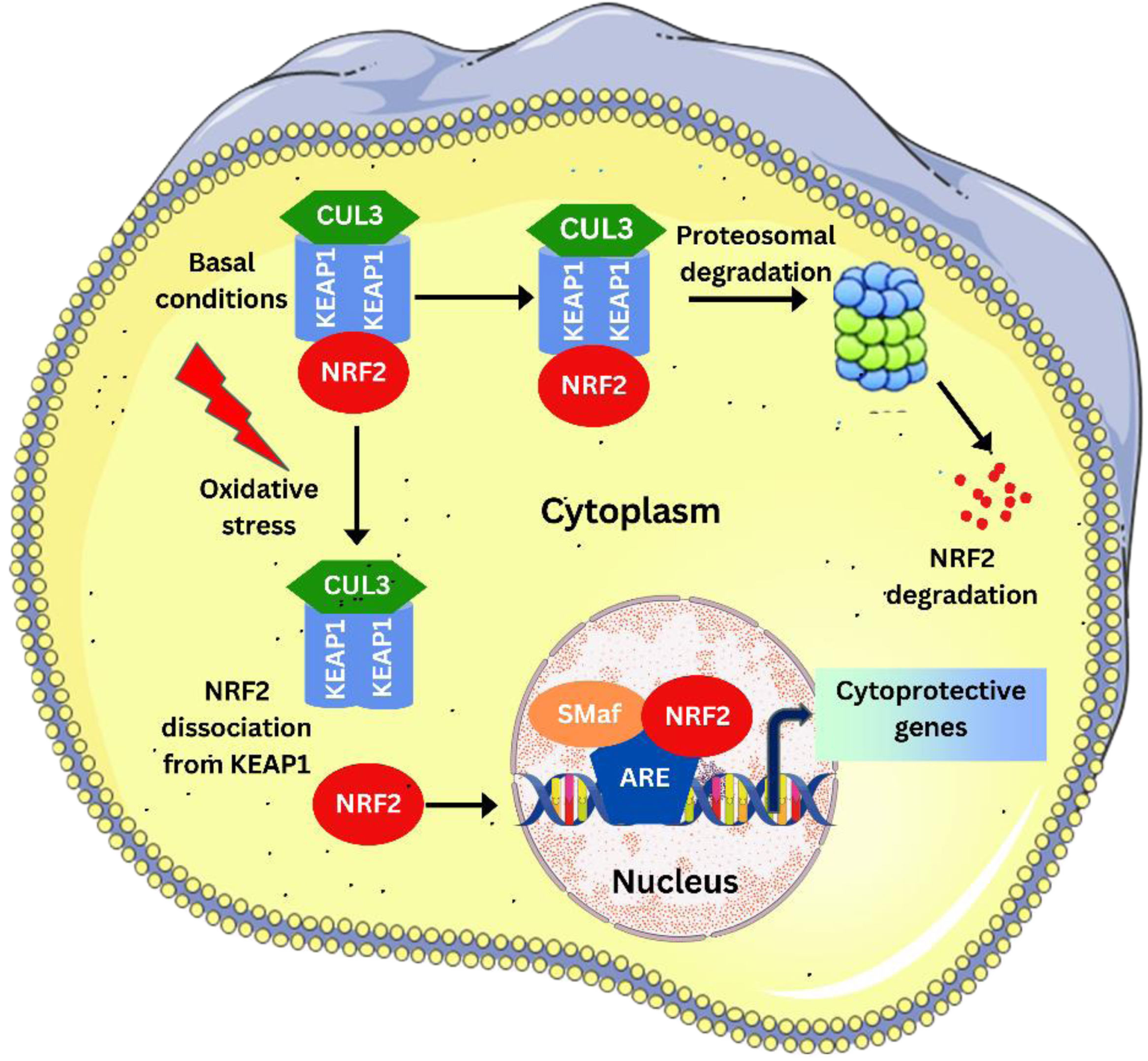
NRF2 signaling pathway
Under basal condition, KEAP1 controlled NRF2 undergoes Cul3 mediated ubiquitination, leading to the proteosomal degradation of NRF2. Under oxidative stress, the NRF2 dissociates from KEAP1 and enters the nucleus where it heterodimerises with the sMaf proteins and regulates the ARE driven gene expression.
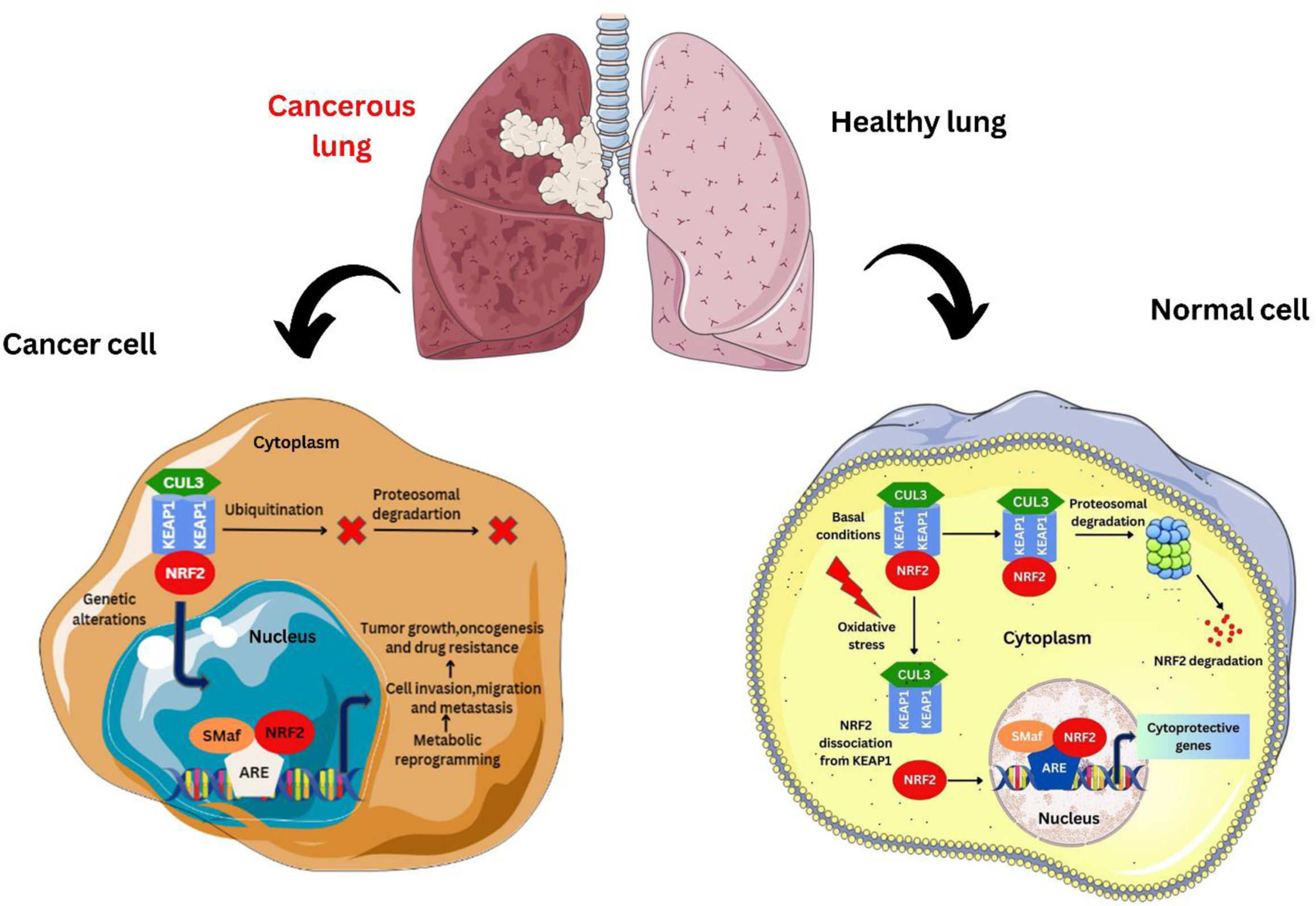
Fate of NRF2 in normal cells and cancer cells.
Loss of KEAP1 function leads to defective activation of the NRF2 signaling pathway in NSCLC cells, resulting in resistance to chemotherapeutic agents.
Omics-To-Therapy (OTT) Team
The Omics-To-Therapy (OTT) Team is at the forefront of translational cancer research, focusing on the intricate molecular networks that govern tumor progression and therapeutic response. Our research leverages multi-omics data—including genomics, transcriptomics, and proteomics—to build a comprehensive understanding of cancer biology. By employing advanced computational biology and AI/ML-driven models, we aim to translate these complex datasets into clinically actionable insights for precision oncology.
A cornerstone of our research is the investigation of the KEAP1-NRF2 signaling pathway, a critical regulator of cellular response to oxidative stress. We have a significant focus on how aberrations in this pathway contribute to the development and progression of cancers, particularly non-small cell lung cancer (NSCLC) and head and neck squamous cell carcinoma.
Beyond the NRF2 pathway, we explore broader themes in cancer research, including sex-based molecular and prognostic disparities in pan-cancer cohorts, the role of immune infiltration in liver cancer, and the identification of multi-omics-based biomarkers for various malignancies. Our ultimate goal is to bridge the gap between fundamental molecular discoveries and their application in the clinic, paving the way for novel therapeutic strategies and improved patient outcomes.